Cohort and case control studies are two types of observational studies used to assess the relationship between exposure and health outcomes.
The investigations are observational rather than experimental because, unlike in a randomized clinical trial, the investigators do not randomly assign patients to exposed or unexposed groups. Confounding is a kind of bias that observational studies are especially vulnerable to since they lack randomization.
Exposures can have protective (like vaccinations) or damaging (like smoking) effects, and the results can include the development of a disease (like lung cancer) or the avoidance of a disease (like infection). Both studies are classified as longitudinal since the study participants are followed for a set period of time.
Cohort Studies
Cohort studies are often conducted prospectively, with the first step being the definition of a population at risk for a certain disease, followed by determining of which cohort members are exposed and which are not exposed.
The number of disease cases for the exposed and unexposed groups is then calculated by following the cohort. In situations where the study period is short (such as during flu season) and follow-up losses are negligible, the probability or risk of developing the relevant disease question is calculated for each group.
The risk among the exposed is calculated by dividing the number of exposed individuals with the outcome of interest by the total number of exposed individuals.
Similarly, the risk among the unexposed is determined by dividing the number of unexposed individuals with the outcome of interest by the total number of unexposed individuals.
In a cohort study, the measure of association is the risk ratio, which is obtained by dividing the risk among the exposed by the risk among the unexposed:
risk ratio = risk in exposed / risk in unexposed
If the probability of disease is higher among the exposed group, then the exposure is considered harmful, and the risk ratio is greater than 1. On the other hand, if the probability of disease is lower among the exposed group, then the exposure is seen as protective, and the risk ratio is less than 1.
In lengthy cohort studies where patients may be lost to follow-up, accurately calculating risks in the exposed and unexposed groups, along with the risk ratio, can become challenging. To address this issue, an alternative measure called the incidence rate is often used.
The incidence rate in the exposed group is calculated by dividing the number of exposed individuals with the outcome of interest by the person-years contributed by all subjects in the exposed group.
Similarly, the incidence rate in the unexposed group is determined by dividing the number of unexposed individuals with the outcome of interest by the person-years contributed by all subjects in the unexposed group.
The measure of association in this scenario is the rate ratio, which is obtained by dividing the incidence rate in the exposed group by the incidence rate in the unexposed group:
rate ratio = incidence rate in exposed / incidence rate in unexposed
Using person-years as the denominator in the incidence rate accounts for the varying lengths of follow-up among study participants and provides a more accurate assessment of the association between exposure and outcome over time.
Doll and Hill’s 1956 cohort study compared the rate at which smokers and non-smokers developed lung cancer.
The study found an incidence rate of 0.84 lung cancer deaths per 1,000 person-years among smokers and 0.07 lung cancer deaths per 1,000 person-years among non-smokers. This results in a rate ratio of 0.84/0.07, which equals 12.
This means that smokers developed lung cancer at a rate 12 times greater than non-smokers. When the rate of disease is higher among the exposed group, such as smokers in this case, the exposure is considered harmful, and the rate ratio is greater than 1.
Conversely, if the rate of disease is lower among the exposed group, the exposure is seen as protective, and the rate ratio is less than 1.
Cohort studies involve two groups that are similar except for their exposure status, recruited from the same source population. Attrition, such as participant loss or dropout, can affect study validity, reducing power and potentially introducing attrition bias – a significant difference between participants who completed the study and those who did not.
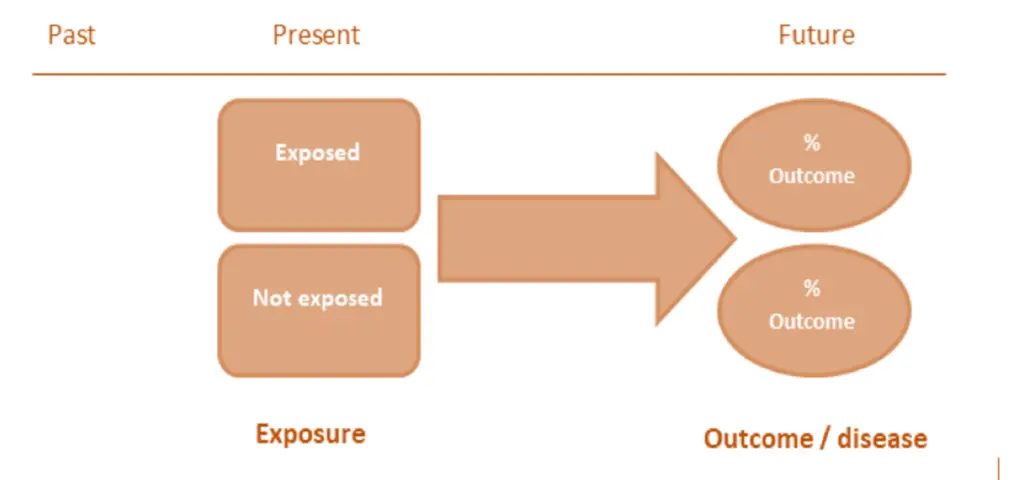
Cohort studies can comprehensively assess various outcomes, allowing for a thorough evaluation of exposure’s impact on disease development. They are particularly beneficial for examining rare exposures, such as exposure to chemical radiation blasts.
Despite their utility, cohort studies can be costly and time-consuming, especially with extended follow-up periods or rare diseases with prolonged latency periods.
Advantages of Cohort Studies
- Enables gathering data on the sequence of events, allowing for causal assessment
- Can assess multiple outcomes related to a specific exposure
- Effective for investigating rare exposures
- Capable of calculating disease rates in exposed and unexposed individuals over time (e.g., incidence, relative risk)
Disadvantages of Cohort Studies
- Requires a large number of subjects when studying rare exposures
- Vulnerable to selection bias
Case control studies
Case control studies are often conducted in a retrospective manner, starting with the identification of people with a certain disease (called cases), followed by the determination of which cases have the relevant exposure.
Next, from the underlying population, a group of subjects (controls) without the disease are selected, and the number of controls with the exposure of interest is determined.
The proportion of subjects with the disease is determined by how the individuals were sampled, not by their probability (risk) of developing the disease, because we sample both subjects with and without the disease.
This means that calculating risks and risk ratios is inaccurate. Instead, the measure of association for a case control study is calculated using an odds ratio.
Case-control studies are retrospective, beginning with two clearly defined groups: one with the disease/outcome and one without. They retrospectively analyze exposure rates to a specific risk factor between these groups to identify potential associations with disease development, although definitive causality cannot be established. The primary outcome measure in case-control studies is the odds ratio (OR).
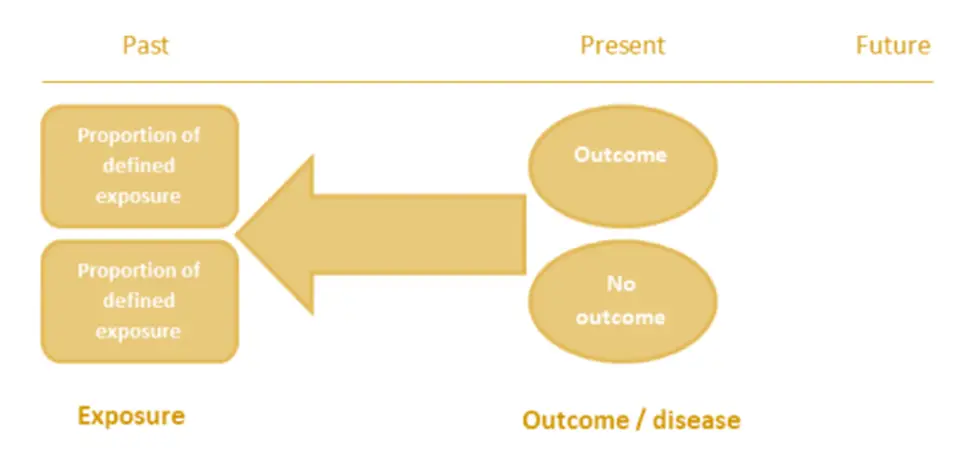
It’s crucial to select cases based on objective criteria from reliable sources like disease registries. However, selecting cases may be challenging due to undiagnosed cases, missed diagnoses, or cases that have died before diagnosis. Controls should be carefully chosen to match cases on relevant factors like age and sex, which can enhance study precision.
Case-control studies offer quick results and are cost-effective, making them suitable for investigating rare diseases or those with long latency periods. They allow the assessment of multiple exposures to understand potential risk factors. However, they are susceptible to biases, particularly recall bias, where differences in recalling past events can affect study outcomes.
To calculate the odds of being exposed among cases and controls, you divide the number of cases exposed by the number of cases unexposed for cases, and divide the number of controls exposed by the number of controls unexposed for controls.
The formula for the odds of exposure among cases is:
odds of exposure among cases = (number of cases exposed) / (number of cases unexposed)
Similarly, for controls:
odds of exposure among controls = (number of controls exposed) / (number of controls unexposed)
Once you have these odds, you can calculate the odds ratio by dividing the odds of exposure among cases by the odds of exposure among controls:
odds ratio = odds of exposure among cases / odds of exposure among controls
This ratio helps assess the association between exposure and outcome, with values greater than 1 indicating a positive association, values less than 1 indicating a negative association, and a value of 1 indicating no association.
Doll and Hill’s 1950 case-control study on smoking and lung cancer revealed that among their cases, there were 688 smokers and 21 non-smokers, resulting in an odds of exposure of 688/21, which equals approximately 32.84. For the controls, there were 650 smokers and 59 non-smokers, leading to an odds of exposure of 650/59, which is approximately 11.0. This yields an odds ratio of 32.84/11.0, which equals approximately 2.98.
This indicates that the odds of smoking among patients with lung cancer are nearly 3 times greater than the odds of smoking among patients without lung cancer. When the odds ratio is greater than 1, as in this case, it means that the odds of exposure are higher among the cases than the controls, suggesting that the exposure (smoking in this study) is harmful.
Conversely, if the odds ratio is less than 1, it indicates that the odds of exposure are lower among the cases than the controls, implying that the exposure could be protective.
Advantages of Case Control Studies
- Suitable for studying rare outcomes or those with long latency
- Quick to conduct compared to other study types
- Cost-effective
- Requires fewer participants
- Utilizes existing records
- Can examine multiple exposures or risk factors
Disadvantages of Case Control Studies
- Prone to recall bias or information bias
- Challenging to validate information
- Control of extraneous variables may be incomplete
- Difficulties in selecting an appropriate comparison group
- Inability to determine disease rates in exposed and unexposed individuals
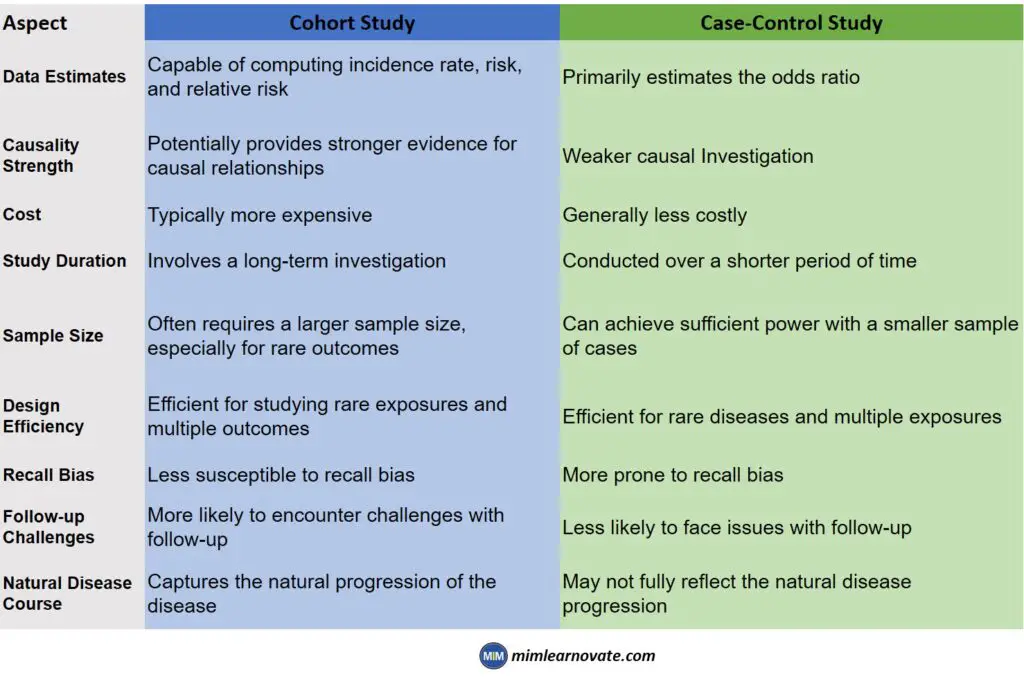
Difference between Cohort Studies and Case Control Studies
| Aspect | Cohort Study | Case-Control Study |
|---|---|---|
| Data Estimates | Capable of computing incidence rate, risk, and relative risk | Primarily estimates the odds ratio |
| Causality Strength | Potentially provides stronger evidence for causal relationships | Weaker causal Investigation |
| Cost | Typically more expensive | Generally less costly |
| Study Duration | Involves a long-term investigation | Conducted over a shorter period of time |
| Sample Size | Often requires a larger sample size, especially for rare outcomes | Can achieve sufficient power with a smaller sample of cases |
| Design Efficiency | Efficient for studying rare exposures and multiple outcomes | Efficient for rare diseases and multiple exposures |
| Recall Bias | Less susceptible to recall bias | More prone to recall bias |
| Follow-up Challenges | More likely to encounter challenges with follow-up | Less likely to face issues with follow-up |
| Natural Disease Course | Captures the natural progression of the disease | May not fully reflect the natural disease progression |
- Cohort studies are usually prospective, requiring careful and detailed follow-up of participants over time. As a result, they tend to be more expensive and time-consuming compared to case-control studies.
- Case-control studies, on the other hand, start with identified cases and are more efficient for studying rare diseases. This efficiency is because enrolling many subjects in a cohort study to ensure some develop a rare disease can be impractical.
- When a disease is rare, the odds ratio in a case-control study can approximate the risk ratio. This approximation is due to the rarity of the disease, which makes the odds of exposure among cases a close estimation of the risk of developing the disease in the exposed group compared to the unexposed group.
A key difference between a case-control study and a cohort study lies in their design and approach:
Cohort studies are more longitudinal in nature, meaning they follow participants over time to observe outcomes. They do not necessarily require a control group, although one can be added if desired by the investigator.
In cohort studies, participants are selected based on a shared characteristic, such as exposure to a risk factor. Retrospective cohort studies specifically look back at a group of people with a common exposure or risk factor and follow them forward in time to observe outcomes.
In contrast, case-control studies require both a case group (individuals with the disease or outcome of interest) and a control group (individuals without the disease).
These studies are designed to identify risk factors for a disease by comparing the characteristics and exposures of cases and controls.
The name “case-control” reflects this design, where cases and controls are compared to understand the association between exposures and the development of the disease.



